Med Quality
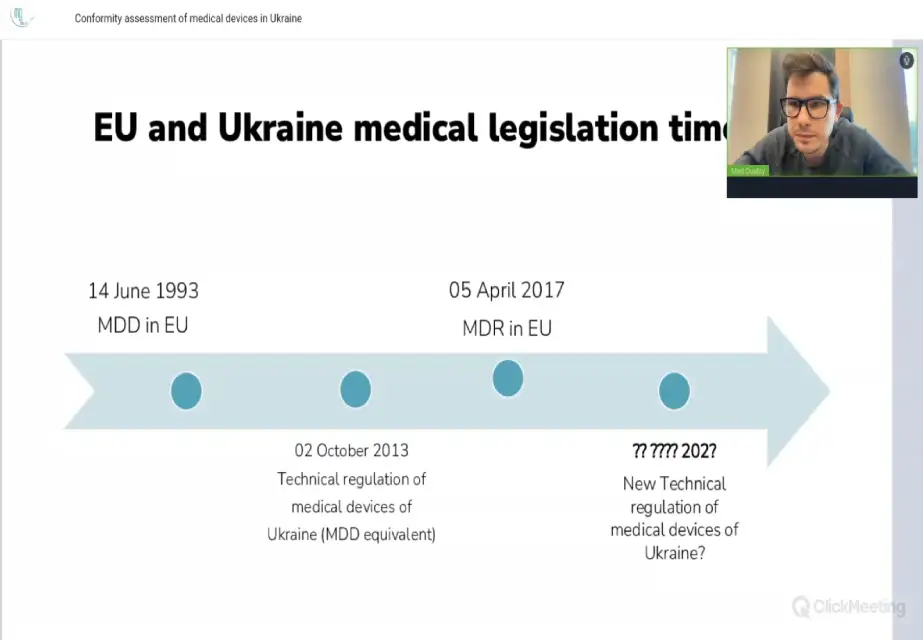
Conformity assessment of medical devices in Ukraine
Qualitas Alta, Qualitas Melior, Qualitas Optima
A leading company providing consulting services for the registration of medical devices and dietary supplements in Ukraine and Europe.
About us
Med Quality
Qualitas Alta, Qualitas Melior, Qualitas Optima
A Ukrainian company providing consulting services for successful conformity assessment and certification for medical devices and dietary supplements. One of our main goals is to assist in access to Ukrainian and European markets while complying with all regulatory requirements. We implement quality management systems according to requirements of ISO 13485 and ISO 22000 standards and will make every effort to get your products to market as quickly as possible. Let’s shape the market for quality products together.
Our services
Complete support during certification process of your medical devices both in conformity assessment body or conducting self-declaration procedure in local authority. This includes communication, document review and preparation, translation, submission of documents to the conformity assessment body and interaction with the State Service of Ukraine on Medicines and Drug Control or Conformity assessment body.
We provide services of authorized representative in Ukraine, comprehensive representation that includes managing customer complaints and support the establishment of a post-market surveillance system for medical devices placed on the market. We streamline all the needed processes and make the market entry as swift as possible.
The implementation of a quality management system according to ISO 13485 requirements ensures that your company meets the highest standards in the medical device field and significantly enhances your rating during tenders. We assist in setting up an effective quality management system that ensures product compliance with the applicable requirements for successful market entry of medical devices.
Full cycle of technical documentation development for medical devices: from risk analysis to organizing and conducting necessary tests. Guarantee of compliance with applicable regulatory requirements and continuous support at all stages of preparing the medical device for market entry.
Identification of potential pathways for medical devices to enter the European market. Document analysis, quality system development, creation and updating of technical documentation to meet the requirements of MDR 2017/745 and IVDR 2017/746 regulations. Market entry planning with clear and transparent stages. Assistance in selecting a notified body within the EU.
Identification of applicable requirements, development of documents for the production of dietary supplements. Translation and proofreading of labeling and instructions for the dietary supplement. Verification of scientific data regarding the effectiveness of the dietary supplement and claims made in the instructions, which will be used in marketing activities.
We conduct the training on the registration and certification of medical devices, ISO 13485, ISO 22000, and other specific applicable standards for medical devices or dietary supplements. Organization and conduction of individual courses where each participant gains not only theoretical knowledge but also practical skills necessary for successful work in the field of medical devices and dietary supplements.
Why choose us?

Team
Our team has gained profound knowledges, skills and expertise in the area necessary for the successful registration of medical devices and dietary supplements.

Individual approach
We understand the unique nature of your product and provide full support at all stages of registration.

Vigiros and result-orientes
We are truelly dedicated to our work and committed to achieving success for our clients.

Reliability
With a commitment to reliability, our consulting services deliver dependable expertise you can count on
Our clients















News
The Director of Med Quality conducted a training on the requirements of ISO 13485:2016 on April 2, 2025.
Med Quality's CEO Vladyslav Kasyanenko hosted webinar on the topic "Conformity assessment of medical devices in Ukraine"
Recently, Med Quality held a webinar on the topic: "How Can Ukrainian Medical Device Manufacturers Enter the European Market? Practical Advice."
Webinars

Conformity assessment of custom-made medical devices: what is really required and what is unnecessary?
26.05.2026

3D printing in medical devices: certification, regulatory requirements, and real risks
23.06.2026

Certification of rehabilitation device components: when do they become a medical device and how to properly classify them? Use of 3D printing in manufacturing
21.07.2026

Labeling of medical devices. Requirements of Technical Regulation No. 753 and ISO 15223-1. How to avoid fines?
28.08.2026

ISO 13485:2016 for manufacturers of rehabilitation aids and other medical devices made to order: what to do with the quality management system?
22.09.2026

Organization and conduct of preclinical testing of prostheses, orthoses, and other rehabilitation devices: from strength testing to biocompatibility
06.10.2026

Post-market surveillance (PMS): how a manufacturer can avoid “failing” after a medical device is placed on the market
20.10.2026

Implant certification: from dentistry to osseointegration, how to properly conduct conformity assessment?
24.11.2026